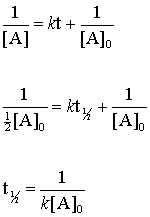half life formula for zero order reaction
T ½ 0693 k For a second order reaction 2A products or A B products when A B rate kA 2. Essentially you can derive them.
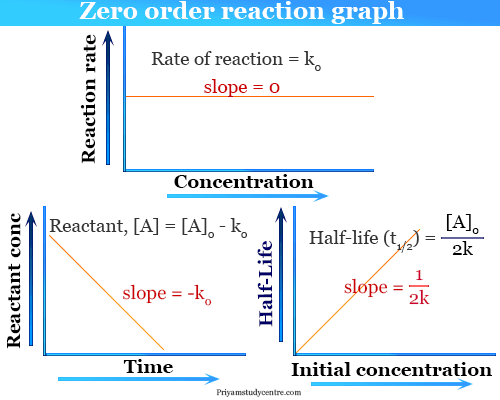
Half life of Zero order reaction formula is the time at which the initial concentration of reactant becomes half and is represented as T 12 C 0 2 k or Half Life of Zero Order Reaction Initial concentration for zero order reaction 2 Rate constant of zero order reaction.

. From the above formula the half-life of the zero order kinetics depends on the initial concentration of the reactant. T ½ 1 k A o Top. NA Product The rate law of zero order kinetics is.
Therefore A2 k 0 t ½ or t ½ A2k. If we set the time t equal to the half-life the corresponding concentration of A at this time is equal to one-half of its initial concentration. Half life formula for nth order reaction.
The half-life of a Zero-th order reaction is t A0 2kHere I derive this from the Integrated Rate LawAsk me questions. Zero Order Half Life Equation - 16 images - half life deranged physiology zero order reaction and its half life chemical kinetics. For a zero order reaction A products rate k.
Determining a half life. The rate constant k will have units of concentrationtime such as Ms due to. The integrated rate constant for the zero-order reaction is given by.
So as the original concentration is decreased the half-life of a 0 th order reaction will also decrease. For a general reaction. Substituting these terms into.
The Method of Half-Lives. Also asked how do you determine the order of a half life reaction. Thus for t t 12 A t ½ A o.
The rate constant for a Zero-order reaction rate of constant k. 1A n-1 1 A 0 n-1 n-1 kt. Here are a number of highest rated Zero Order Half Life Equation pictures on internet.
Equations for Half Lives. Its submitted by dealing out in the best field. Half life means 50 percent of reactants disappear in that time interval.
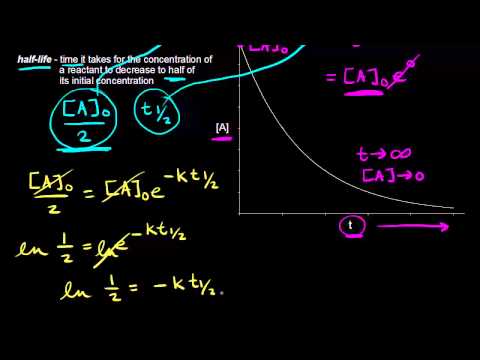
The half-life of a reaction is defined as the time required for the reactant concentration to fall to one half of its initial value. This depicts a zero-order reaction. Half life in zero order reaction.
The Initial concentration for zero order reaction is the concentration of reactant present before the start. If we know the integrated rate laws we can determine the half-lives for first- second- and zero-order reactions. Solving for t12 reveals the dependence of the half-life on the initial concentration.
The Half-Life of Zero Order Reaction calculator computes the half-life in nuclear decay for a zero order reaction. A zero order reaction implies that the rate of the reaction does not depend on the concentration of the reactant. We can derive an equation for determining the half-life of a first-order reaction from the alternate form of the integrated rate law as follows.
For the first-order reaction the half-life is defined as t 12 0693k. Converting a half life to a rate constant. The rate constant for a zero-order reaction is measured in molL-1s-1.
Postby Acharya Ranawat 3E Sun Mar 13 2022 221 am. The mathematical expression that can be employed to determine the half-life for a zero-order reaction is t 12 R 0 2k. The half-life is the time required for a quantity to fall to half its initial value as measured at the beginning of the time period.
Zero-Order kinetics describe reactions and processes where the rate and half-life are. T ½ A o 2k For a first order reaction A products rate kA. As for other reaction orders an equation for zero-order.
When t t ½ that is the half-life of the reaction completed the concentration of the reactant A A2. T12 A 02K. We know that at the half-life time eqt_12 eq the concentration of the reactant will.
Using the concentration-time equation for a second-order reaction we can solve for half-life. Half-Life of Zero Order Reaction. It is to be noted that the half-life of a zero-order reaction is determined by the initial concentration and rate constant.
5 rows Zero-Order Reactions. Fri Sep 24 2021 139 pm. Graphical relations and half lives.
Looking at the equation for zero order A-ktA0 if we make A12A0 we get that tA02k. This is an expression of the half-life of a zero-order reaction. We identified it from obedient source.
And for the second-order reaction the formula for the half-life of. The half-life of a zero-order reaction the formula is given as t 12 R 02 k The half-life of a first-order reaction is given as t 12 0693k The half-life of a second-order reaction is given by the formula 1kR 0. Half life for 0 and 2nd order reactions.
Where A 0 Initial concentration of reactant at timet 0.
Half Life Of A First Order Reaction Video Khan Academy

Half Life Of A Third 3rd Order Reaction Youtube

Half Life Of Zero Th 0th Order Reaction Derivation Youtube
Zero Order Reaction Definition Examples Formula

Zero Order Reactions Video Kinetics Khan Academy

Shelf Life Of Foods First Order Kinetics Youtube

Csir Ugc Net Half Life Of Zero Order Reaction In Hindi Offered By Unacademy

Summary Of The Kinetics Of Zero Order First Order Ppt Download

Half Life Expressions Chemistnate

Zero Order Reactions Chemistry Class 12 Iit Jee Main Advanced Neet Aipmt Askiitians Youtube

Rate Of Zero Order Reaction Integrated Law Half Life Period Rate Constant

Half Life Expressions Chemistnate

Principles And Kinetics Of Drug Stability Phr 416 Ppt Video Online Download